- What is a standard drink? National Institute on Alcohol Abuse and Alcoholism
- 1998, What is a “standard drink”? International Center for Alcohol Policies
- Bober B et al, Alcohol diffusion in the stomach lining The University California, San Diego
- Klockhoff H et al, 2002, Faster absorption of ethanol and higher peak concentration in women after gastric bypass surgery PubMed Central
- Kechagias S et al, 1997, Low-dose aspirin decreases blood alcohol concentrations by delaying gastric emptying PubMed
- Alcohol Absorption The National Academic Press
- Smoking alcohol — a dangerous re-immerging trend Iowa.gov
- Why you can’t fool quality breath testing instruments? Lion Laboratories
- Lachenmeier DW, 2008, Safety evaluation of topical applications of ethanol on the skin and inside the oral cavity PubMed Central
- Hansen CS et al, 2010, Testing the validity of the Danish urban myth that alcohol can be absorbed through feet: open labelled self experimental study PubMed Central
- Paton A, 2005, Alcohol in the body The BMJ
- Fenna D et al, 1971, Ethanol metabolism in various racial groups PubMed Central
- Ye L, 2009, Alcohol and the Asian flush reaction University of Guelph
- Silver SQ et al, 1999, De novo lipogenesis, lipid kinetics, and whole-body lipid balances in humans after acute alcohol consumption The American Journal of Clinical Nutrition
- Elia M et al, 2007, Energy values of macronutrients and specific carbohydrates in foods European Journal of Clinical Nutrition
- Bullock C, 1990, THE BIOCHEMISTRY OF ALCOHOL METABOLISM- A BRIEF REVIEW Wiley Online Library
- Alcohol absorption, distribution & elimination Forcon.ca
- Levitt DG et al, 2002, PKQuest: measurement of intestinal absorption and first pass metabolism – application to human ethanol pharmacokinetics BMC Pharmacology and Toxicology
- Levitt MD et al, 1997, Use of measurements of ethanol absorption from stomach and intestine to assess human ethanol metabolism Gastrointestinal and Liver Physiology
- Brown AS et al, 1995, The effect of gastritis on human gastric alcohol dehydrogenase activity and ethanol metabolism PubMed
- Barceloux DG, 2012, p.312, Medical Toxicology of Drug Abuse: Synthesized Chemicals and Psychoactive Plants
- Dettling A et al, 2007, Ethanol elimination rates in men and women in consideration of the calculated liver weight PubMed
- Jones AW, 2010, Evidence-based survey of the elimination rates of ethanol from blood with applications in forensic casework PubMed
- Mizoi Y et al, 1987, Individual and ethnic differences in ethanol elimination PubMed
- Mascord D et al, 1991, THE EFFECT OF FRUCTOSE ON ALCOHOL METABOLISM AND ON THE [LACTATE]/[PYRUVATE] RATIO IN MAN Alcohol and Alcoholism
- Soterakis J et al, 1975, Increased rate of alcohol removal from blood with oral fructose and sucrose The American Journal f Clinical Nutrition
- Uzuegbu UE et al, 2009, Fructose-induced increase in ethanol metabolism and the risk of Syndrome X in man PubMed
- Crownover BP et al, 1986, Activation of ethanol metabolism in humans by fructose: importance of experimental design The Journal of Pharmacology and Experimental Therapeutics
- Berman PAM et al, 2003, Effect of oral fructose on ethanol elimination from the bloodstream Refdoc.fr
- 1995, Carbohydrates and the Sweetness of Honey Honey.com
- Levy R et al, 1977, Intravenous fructose treatment of acute alcohol intoxication. Effects on alcohol metabolism PubMed
- Pavlic M et al, 2007, Another ‘soberade’ on the market: does Outox keep its promise? PubMed
- Begleiter H, The Pharmacology of Alcohol and Alcohol Dependence
- Lisander B et al, 2005, ENHANCED RATE OF ETHANOL ELIMINATION FROM BLOOD AFTER INTRAVENOUS ADMINISTRATION OF AMINO ACIDS COMPARED WITH EQUICALORIC GLUCOSE Alcohol and Alcoholism
- Oneta CM et al, 1998, First pass metabolism of ethanol is strikingly influenced by the speed of gastric emptying Gut
Alcohol Absorption, Metabolism and Elimination Rate
Terms Explanation
ABV
% abv (percent of alcohol by volume) is the volume percent of alcohol in an alcoholic beverage.
A “Standard Drink”
In the United States and Canada, one standard drink contains about 14 grams of alcohol, which is 0.6 fl. oz or 18 milliliters of pure alcohol [1].
One standard drink is:
- 12 oz (355 mL) of 5% beer
- 8 oz (240 mL) of 7.5% malt liquor
- 5 oz (150 mL) of 12% wine
- 3.5 oz (105 mL) of 17% fortified wine
- 2.5 oz (75 mL) of 24% liqueur
- 1.5 oz (45 mL), a jigger or a “shot” of 80 proof (40%) distilled alcohols (gin, rum, vodka, tequila, whiskey)
One 12 oz can of 5% beer contains about the same amount of alcohol as one jigger of 40% whiskey, so it is misleading to say that beer contains less alcohol than whiskey.
Picture 1. Examples of a standard drink
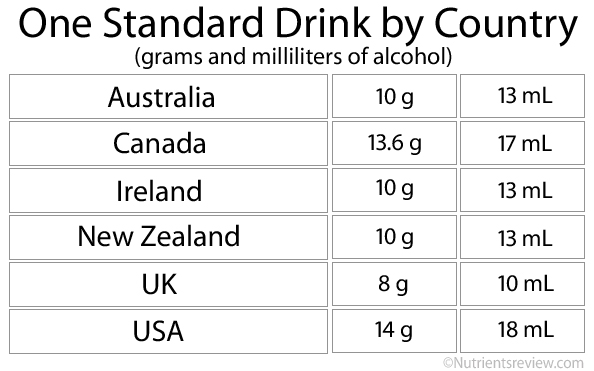
Picture 2. A standard drink in various countries [2]
Alcohol Absorption
When you drink alcohol on an empty stomach, about 20% of it will be absorbed in the stomach and about 80% in the small intestine [3]. Minute amounts can be absorbed in the mouth [3]. Ethanol does not need to be digested (broken down) in order to be absorbed [8].
Alcohol absorption rate increases with:
- Alcohol percent of an alcoholic beverage, but only up to about 30% abv.
- Factors that stimulate stomach emptying:
- Drugs: erythromycin, ranitidine [35]
- Gastric bypass surgery [4]
Alcohol absorption rate decreases with:
- Factors that slow stomach emptying: food, drugs, such as narcotics and aspirin [5], sugar and fat in alcoholic beverages.
Alcohol Absorption After Inhalation
Inhaling alcohol from the air in bars and alcohol-producing factories does not likely result in intoxicating levels of alcohol in the blood [8]. In one study, participants after inhaling alcohol from the alcohol-saturated air, had their maximal blood alcohol concentrations below 0.005 g/100 mL blood and no intoxication symptoms [6].
Inhaling concentrated alcohol vapor through a nebulizer device AWOL (Alcohol WithOut Liquid) may result in intoxicating blood alcohol levels [7].
Alcohol Absorption Through the Skin and Mouth
Absorption of alcohol through the intact skin in an adult, for example, by using alcohol hand sanitizers [8,9] or by submerging the feet in alcoholic beverages [10], does not result in intoxicating blood alcohol levels. Theoretically, using large amount of alcohol to disinfect an injured skin in a child could result in intoxication blood alcohol levels [9].
Rinsing the mouth with alcohol-containing mouthwash does not likely result in intoxicating blood alcohol levels [9].
Alcohol Distribution in the Body
From the gut alcohol is absorbed into the blood from where it is distributed into the body tissues. Alcohol is more soluble in water than in fat, so it will readily enter the muscles (which are high in water) but not the fat tissues, which means that individuals with high body fat percent will have more alcohol in the blood than the lean ones of the same body weight after drinking the same amount of alcohol [11].
How is alcohol metabolized?
After absorption, more than 90% of ethanol is metabolized in the liver [16]. One gram of ethanol provides about 7 Calories [15].
Chart 1. Alcohol Metabolism |
|
| Three steps of ethanol metabolism: | Factors that may affect the rate of ethanol metabolism: |
| 1. Ethanol is converted, by the enzyme alcohol dehydrogenase (ADH), into acetaldehyde. |
|
| 2. Acetaldehyde is converted, by the enzyme aldehyde dehydrogenases (ALDH), into acetate. | In many East Asians, the enzyme ALDH is less active, so it cannot efficiently produce acetate from acetaldehyde, so the later accumulates in their blood and cause facial flushing [13]. |
| 3. Acetate is partly converted into fatty acids, but mainly broken down into carbon dioxide and water. | Acetate is broken down and used as a source of energy [14]. Acetate in excess of calorie needs is converted to body fat [14]. |
Alcohol Elimination From the Body
More than 90% of the absorbed alcohol is broken down and eliminated by the liver, up to 5% is removed from the body by breath, up to 5% by urine, and only negligible amounts by the stool and sweat [17]. The breakdown of alcohol by the enzyme gastric alcohol dehydrogenase in the stomach is probably negligible [18,19,20].
The alcohol elimination rate is pretty constant in a certain individual in the same circumstances and may range from about 0.010 to 0.060 grams of alcohol/100 ml blood/hour among individuals [21; p.387]. The average alcohol elimination rate in mild and moderate drinkers is estimated to 0.015 g/100 mL blood/hour [23].
The AVERAGE alcohol elimination rates in different circumstances [23]:
- Moderate drinkers drinking on an empty stomach: 0.010-0.015 grams alcohol/100 milliliters blood/hour.
- Moderate drinkers drinking after a meal: 0.015-0.020 g/100 mL/hour. A solid meal containing about 550 kilocalories, regardless of its composition (carbohydrates, proteins, fats) eaten one hour before injection of alcohol into a vein (resulting in BAC 0.05 g/100 mL) can speed up the alcohol elimination by 25-45% [21; p.386]. The probable mechanism is a food-induced increased blood flow to the liver and thus increased delivery of alcohol to the liver. Even eating several hours after drinking may speed up alcohol elimination by up to 50%.
- Drivers arrested from “drunk driving,” who are often binge drinkers: 0.020 g/100 mL/hour.
- Chronic alcoholics: 0.025-0.035 g/100 mL/hour (but up to 0.062 g/100 mL/h has been observed) [21; p.387]. Alcohol elimination rate increases only after several weeks of heavy drinking; in one study, alcohol elimination rate in non-alcoholics did not increase after drinking about 3 drinks per day for 3 weeks [21; p.387].
What else can affect alcohol elimination rate?
INCREASED alcohol elimination rate:
Women may have slightly higher alcohol elimination rate (up to about 20%) than men, possibly due to greater average liver/body weight ratio in women; menstrual cycle does not significantly affect the elimination rate [21-p.386;22].
Ingestion of 60-150 grams fructose after drinking alcohol can, according to several studies, speed up alcohol elimination by 20-80% [25,26,27,28,29]. For example, in one study, participants who have been drinking alcohol and reached BAC about 0.1 g alcohol/100 mL blood, and then ingested 1 gram fructose/kg body weight one hour after drinking reached zero BAC 90 minutes earlier than when drinking without fructose [28]. According to another study, fructose taken after drinking could shorten the time in which the blood alcohol concentration in a severely intoxicated person (0.25 g/100 mL) would drop to a “sober” level (0.05 g/100 mL) by up to four hours (9 hours with fructose vs. 13 hours without fructose) [26]. Giving fructose to a person who has a liver disease and is acutely poisoned with alcohol could aggravate the symptoms of poisoning [26]. To get 100 g fructose, you would need to drink about 2 liters of HFCS-sweetened soda or eat about 300 g of honey, 500 g of prunes or 300 g of dried apples, which could easily make you sick or cause diarrhea, though [30]. In some studies, fructose consumption had no significant effect on the alcohol elimination rate, though [31,32].
Intravenous infusion of amino acids may speed up alcohol elimination by about 60% [34].
DECREASED alcohol elimination rate:
Individuals with ALDH2 deficiency (mostly East Asians) have lower alcohol elimination rate than healthy individuals [24].
Shock, for example, after a car crash, may slow down alcohol elimination by about 50%; this may greatly increase the estimated amount of blood alcohol concentration at the time of the accident [33].
What does NOT significantly affect the alcohol elimination rate?
- Age does not significantly affect the alcohol elimination rate, but children may eliminate alcohol at the rate up to 0.028 g/100 mL blood/hour, probably due to greater liver/body weight ratio [21; p.386].
- “Sobriety drinks” or “soberades” containing fructose do not seem to significantly stimulate alcohol elimination from the body, probably due to relatively low fructose amount in a 250 mL bottle [32].
- There seems to be no evidence that drinking large amounts of water [21; p.386], taking caffeine, salt (sodium) or contraceptive pills or smoking would significantly affect alcohol elimination.
- Drugs: indoramin (alpha-blocker), propranolol (beta-blocker), cefpirome (antibiotic), aspirin, acetaminophen, cimetidine, omeprazole probably do not significantly affect alcohol elimination [21- p.388; 33].
Alcohol
- Alcohol chemical and physical properties
- Alcoholic beverages types (beer, wine, spirits)
- Denatured alcohol
- Alcohol absorption, metabolism, elimination
- Alcohol and body temperature
- Alcohol and the skin
- Alcohol, appetite and digestion
- Neurological effects of alcohol
- Alcohol, hormones and neurotransmitters
- Alcohol and pain
- Alcohol, blood pressure, heart disease and stroke
- Women, pregnancy, children and alcohol
- Alcohol tolerance
- Alcohol, blood glucose and diabetes
- Alcohol intolerance, allergy and headache
- Alcohol and psychological disorders
- Alcohol and vitamin, mineral and protein deficiency
- Alcohol-drug interactions
- Fructose
- Galactose
- Glucose
- Isomaltose
- Isomaltulose
- Lactose
- Maltose
- Mannose
- Sucrose
- Tagatose
- Trehalose
- Trehalulose
- Xylose
- Erythritol
- Glycerol
- Hydrogenated starch hydrolysates (HSH)
- Inositol
- Isomalt
- Lactitol
- Maltitol
- Mannitol
- Sorbitol
- Xylitol
- Fructo-oligosaccharides (FOS)
- Galacto-oligosaccharides (GOS)
- Human milk oligosaccharides (HMO)
- Isomalto-oligosaccharides (IMO)
- Maltotriose
- Mannan oligosaccharides (MOS)
- Raffinose, stachyose, verbascose
- SOLUBLE FIBER:
- Acacia (arabic) gum
- Agar-agar
- Algin-alginate
- Arabynoxylan
- Beta-glucan
- Beta mannan
- Carageenan gum
- Carob or locust bean gum
- Fenugreek gum
- Galactomannans
- Gellan gum
- Glucomannan or konjac gum
- Guar gum
- Hemicellulose
- Inulin
- Karaya gum
- Pectin
- Polydextrose
- Psyllium husk mucilage
- Resistant starches
- Tara gum
- Tragacanth gum
- Xanthan gum
- INSOLUBLE FIBER:
- Cellulose
- Chitin and chitosan
- FATTY ACIDS
- Saturated
- Monounsaturated
- Polyunsaturated
- Short-chain fatty acids (SCFAs)
- Medium-chain fatty acids (MCFAs)
- Long-chain fatty acids (LCFAs)
- Very long-chain fatty acids (VLCFAs)
- Monoglycerides
- Diglycerides
- Triglycerides
- Vitamin A - Retinol and retinal
- Vitamin B1 - Thiamine
- Vitamin B2 - Riboflavin
- Vitamin B3 - Niacin
- Vitamin B5 - Pantothenic acid
- Vitamin B6 - Pyridoxine
- Vitamin B7 - Biotin
- Vitamin B9 - Folic acid
- Vitamin B12 - Cobalamin
- Choline
- Vitamin C - Ascorbic acid
- Vitamin D - Ergocalciferol and cholecalciferol
- Vitamin E - Tocopherol
- Vitamin K - Phylloquinone
- Curcumin
- FLAVONOIDS:
- Anthocyanidins
- Flavanols: Proanthocyanidins
- Flavanones: Hesperidin
- Flavonols: Quercetin
- Flavones: Diosmin, Luteolin
- Isoflavones: daidzein, genistein
- Caffeic acid
- Chlorogenic acid
- Lignans
- Resveratrol
- Tannins
- Tannic acid


4 Responses to "Alcohol Absorption, Metabolism and Elimination Rate"